Abstract
Albeit novel treatment concepts have entered the therapeutic landscape of AML, relapse rates remain high. This is attributed to treatment-refractory leukemic cells, which can be quantified by flow cytometry and molecular techniques (Measurable Residual Disease, MRD). MRD positivity is a significant prognostic biomarker for relapse and low overall survival rates. Post-remission therapy for the elimination of MRD remains a major challenge. We previously confirmed the prognostic value of early flow MRD assessment during aplasia in patients who achieve complete remission after intensive induction therapy (Koehnke et al., 2019). Here, we hypothesized that single-cell transcriptional profiling of refractory leukemic cells using flow cytometry (flow MRD) during aplasia will help to understand resistance mechanisms to therapy and to identify suitable targets for cellular immunotherapy.
Flow MRD detection is based on the definition and characterization of leukemia-associated aberrant immunophenotypes (LAIPs) at the time of initial diagnosis, as described previously (Koehnke et al., 2015). A similar approach was applied to define LAIP+ cells at the time of aplasia (d16-d21 after initiation of induction therapy). For single-cell RNA-seq, LAIP+ cells from paired initial diagnosis and aplasia were sorted and subjected to transcriptional profiling using the mcSCRB-seq (molecular crowding single-cell RNA barcoding and sequencing) protocol (Bagnoli et al., 2018).
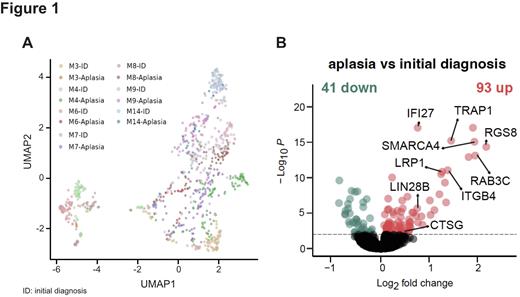
We included 7 AML patients of different ELN genetic risk group (favorable, n=1; intermediate, n=4; adverse, n=2). In total, 854 LAIP+ cells from paired initial diagnosis-aplasia samples were projected using uniform manifold approximation and projection (UMAP) visualizations. We observed a distinct clustering of LAIP+ cells per patient and time point, revealing high inter-patient heterogeneity (Figure 1A). To identify common transcriptional changes across patients, we performed differential gene expression analysis of LAIP+ cells, comparing aplasia to initial diagnosis. We identified 93 genes significantly upregulated and 41 genes significantly downregulated (p < 0.01, FC >1 or <-1; Figure 1B). Among markedly upregulated genes across patients at the time of aplasia, we found 3 genes related to leukemic cell maintenance. Interestingly, SMARCA4, an ATPase subunit of the SWI/SNF chromatin remodeling complex, known to be essential for the proliferation and survival of AML cells (Buscarlet et al., 2014; Shi et al., 2013), showed a 1.99-fold increase in expression (p < 0.00). In addition, TRAP1, a candidate target in cancers with antiapoptotic function, was upregulated 1.5-fold (p < 0.00). And lastly, LIN28B, the cancer stem cell and leukemia driver gene, was also found to be upregulated by 0.7-fold in aplasia LAIP+ cells (p < 0.00). In addition, several immune-related genes including DC marker gene, LRP1 (CD91; p < 0.00, FC = 1.3) and the neutrophil/GMP- specific gene, cathepsin G (CTSG; p < 0.01, FC = 0.5) showed a significant up-regulation in aplasia LAIP+ cells. The latter was previously identified as a leukemia-associated antigen and a potential target for immunotherapy (Alatrash et al., 2017). Strikingly, LAIP+ cells at the time of aplasia harbored a LSC gene expression pattern in a patient-specific manner. Lastly, cell type prediction using the annotated dataset (van Galen et al., 2019) assigned the phenotype of LAIP+ cells mainly to GMP-like and cDC-like cells in variable abundance across patients.
In summary, our data showed high inter-patient heterogeneity and phenotypic differences, making general statements on chemo-resistance challenging. However, despite this heterogeneity, LAIP+ cells at the time of aplasia harbor remarkable proliferation and survival advantage (stem cell-like phenotype), specifically, due to the upregulation of SMARCA4, TRAP1, and LIN28B genes. This phenotype is accompanied by patient-specific enrichment of LSC signature and upregulation of immune-related genes. Our data open up the path to better understand chemo-resistance and to identify targets that need to be addressed to eradicate MRD and improve relapse-free survival.
Disclosures
Heitmüller:Morphosys: Current Employment. Koehnke:TenSixteen Bio: Consultancy. Schnorfeil:Bavarian Nordic GmbH: Current Employment; PPD Germany GmbH & Co KG: Ended employment in the past 24 months; Medigene Immunotherapies GmbH: Ended employment in the past 24 months. Metzeler:Astellas: Honoraria; Pfizer: Consultancy; AbbVie: Honoraria; Celgene/BMS: Consultancy, Honoraria, Research Funding; Daiichi Sankyo: Honoraria; Novartis: Consultancy; Jazz Pharmaceuticals: Consultancy. Hopfner:Boehringer Ingelheim: Research Funding. Buecklein:Miltenyi Biotech: Research Funding; Novartis: Honoraria, Research Funding, Speakers Bureau; Pfizer: Research Funding, Speakers Bureau; Amgen: Honoraria; Gilead: Honoraria. Subklewe:Miltenyi Biotech: Research Funding; Janssen: Consultancy, Speakers Bureau; Seagen: Research Funding; Takeda: Other: Travel support; Morphosys: Research Funding; Novartis: Consultancy, Speakers Bureau; Roche: Consultancy, Research Funding; Gilead: Consultancy, Research Funding, Speakers Bureau; Celgene/BMS: Consultancy, Speakers Bureau; Amgen: Consultancy, Research Funding, Speakers Bureau; Pfizer: Consultancy; Seattle Genetics: Research Funding; Bristol-Myers Squibb: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal